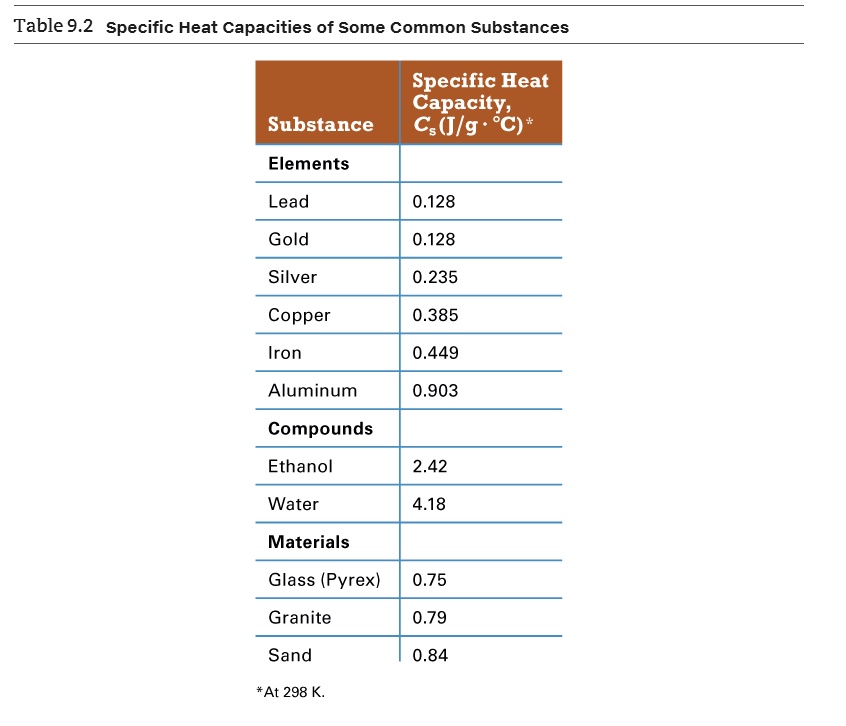
SOLVED: Table 9.2 Specific Heat Capacities of Some Common Substances Specific Heat Capacity; Cs(Jlg. *C) Substance Elements Lead 0.128 Gold 0.128 Silver 0.235 Copper 0.385 Iron 0.449 Aluminum 0.903 Compounds Ethanol 2.42

Specific heat and magnetization studies of spin-glass like transition in nanogranular Cu90Co10 ribbon - ScienceDirect

Specific heat capacities for an as-deposited thin toluene film (solid... | Download Scientific Diagram

SOLVED: Aheat energy of 645 J is applied to a sample of glass with a mass of 28.4g: point Its temperature increases from -11.6 'C to 15.5 'C. Calculate the specific heat
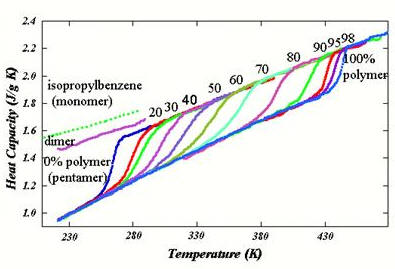
Thermodynamics of The Glass Transition | Research Areas | Polymers and Condensed Matter Physics Group | Research | Department of Chemical Engineering | TTU

Heat capacity (C p ) vs. temperature curves in the glass transition... | Download Scientific Diagram



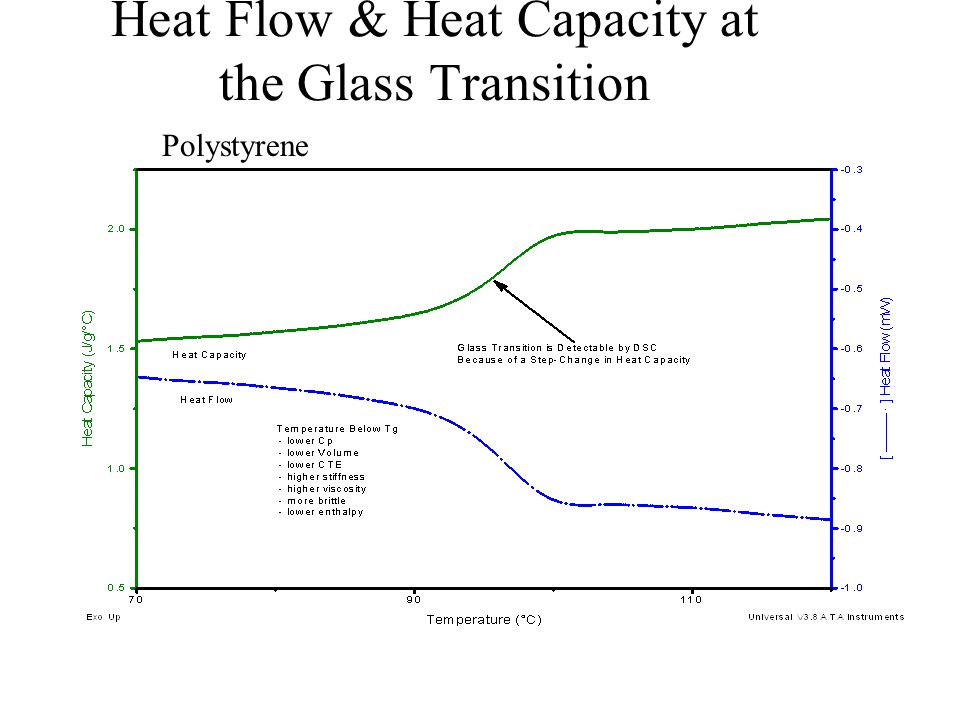

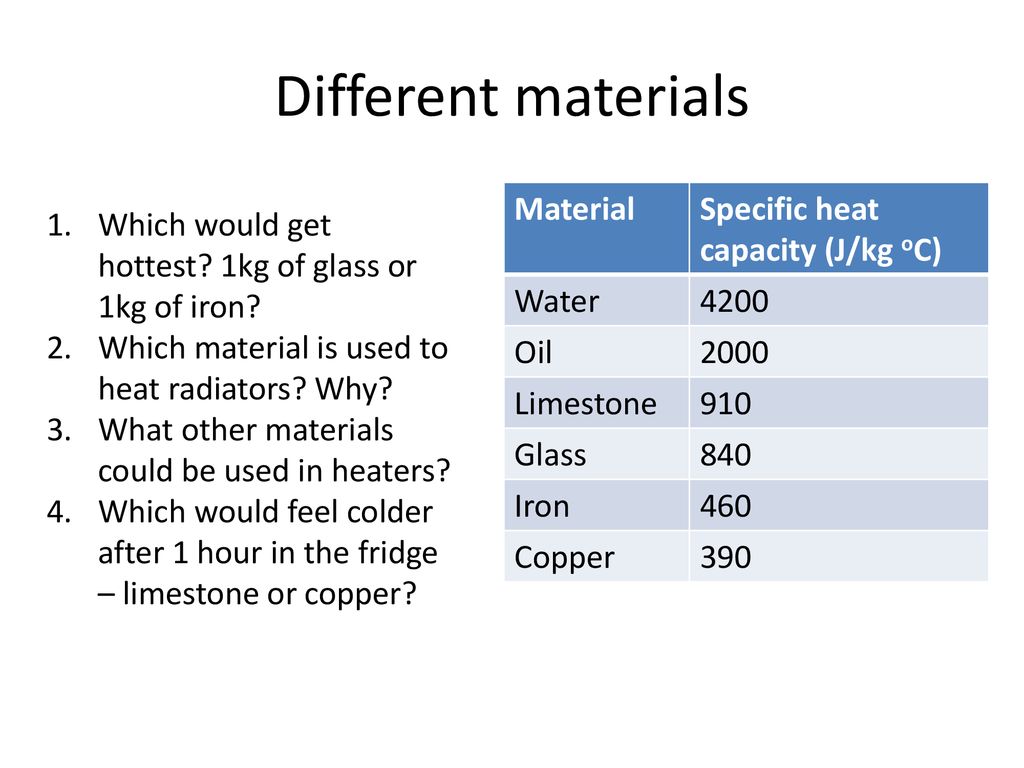



.jpg)
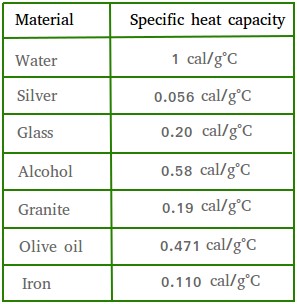
.jpg)