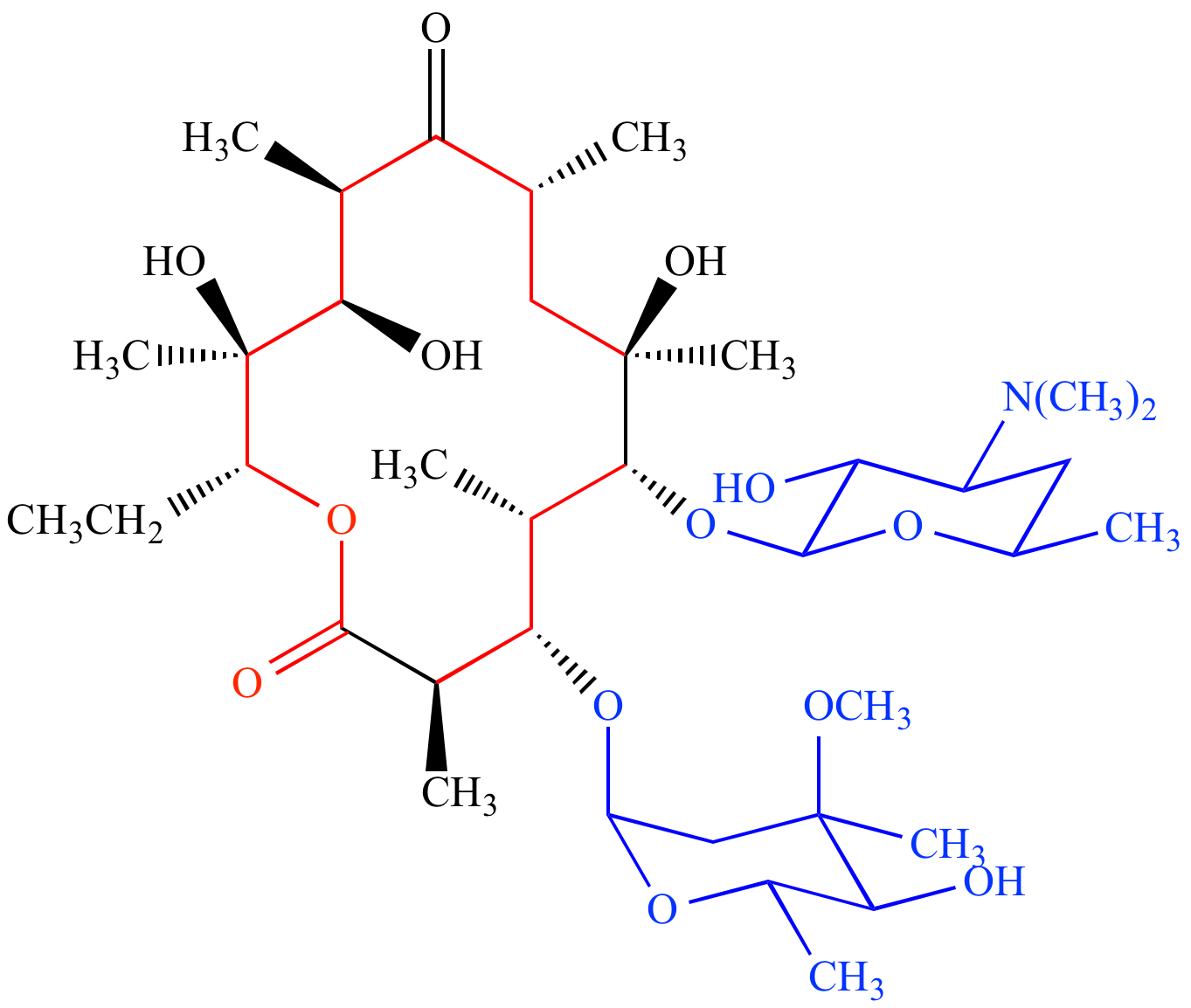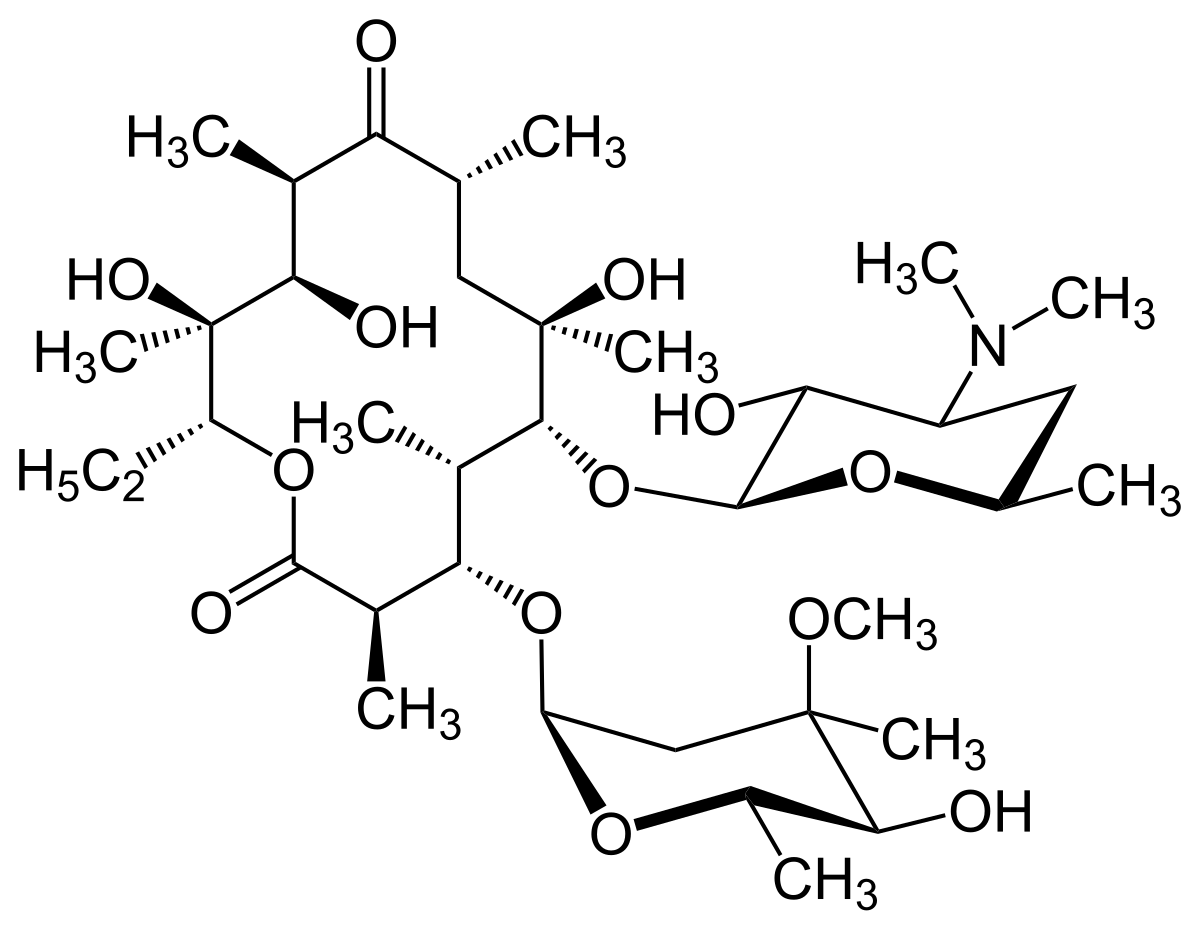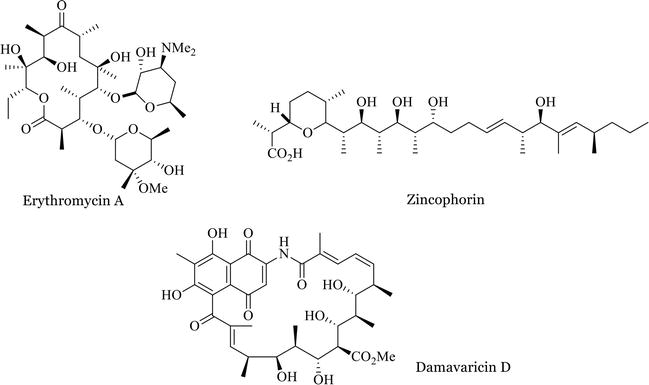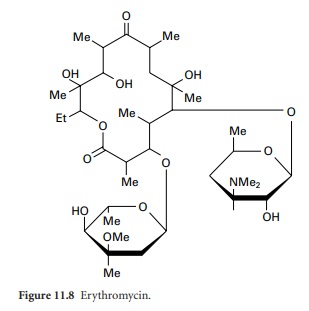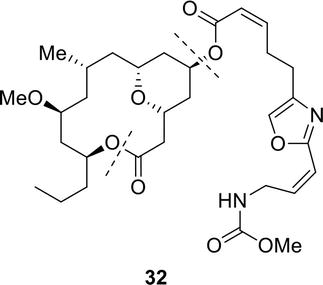
The actinomycetes. Actinomycetales. CHEMICAL NATURE OF ANTIBIOTICS 55 the data pul)lished on the various polyenes can he found in Part 1^ of this book. One should keep in mind that many

Synthesis of New 14-Membered Macrolide Antibiotics via a Novel Ring Contraction Metathesis | Organic Letters
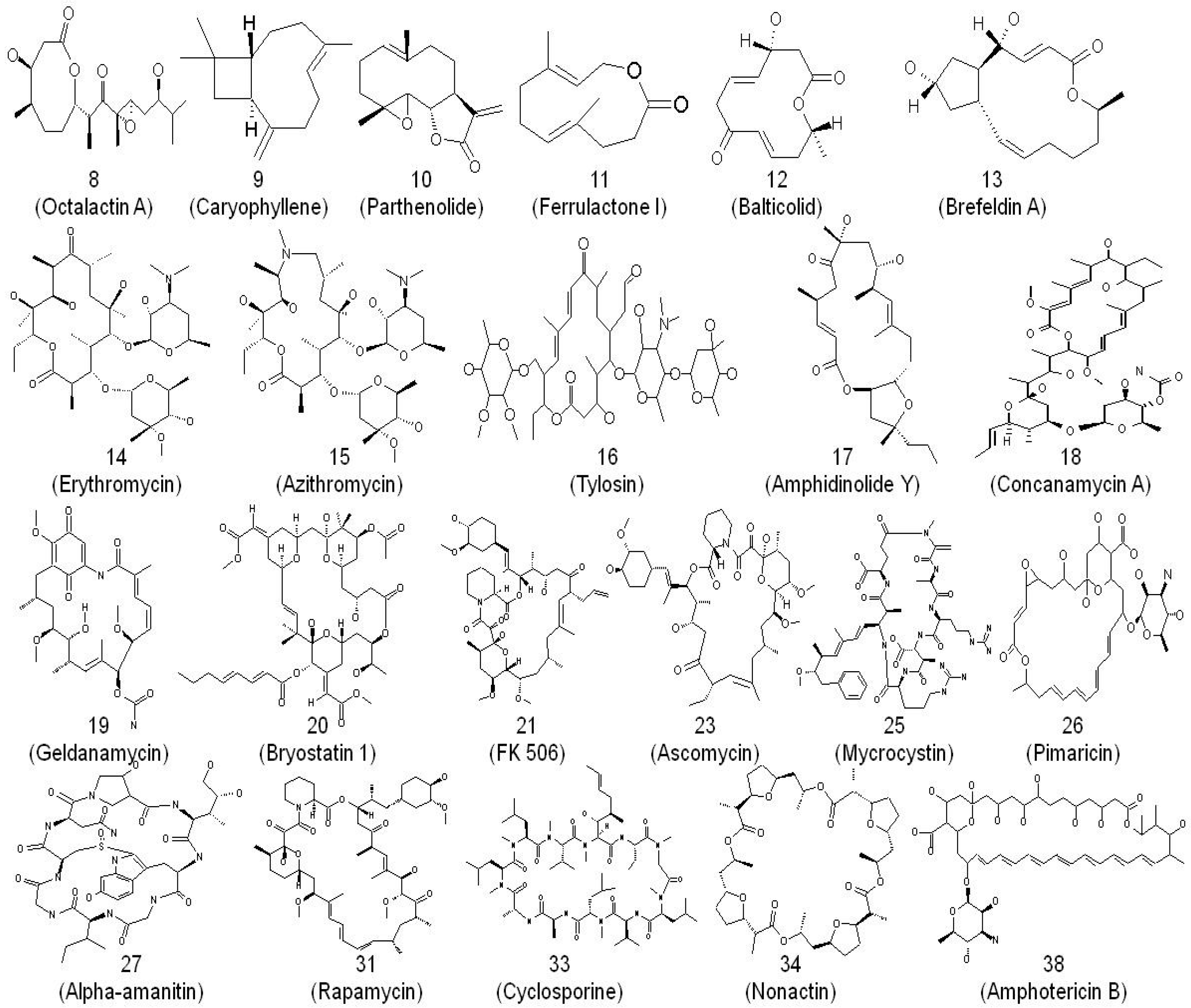
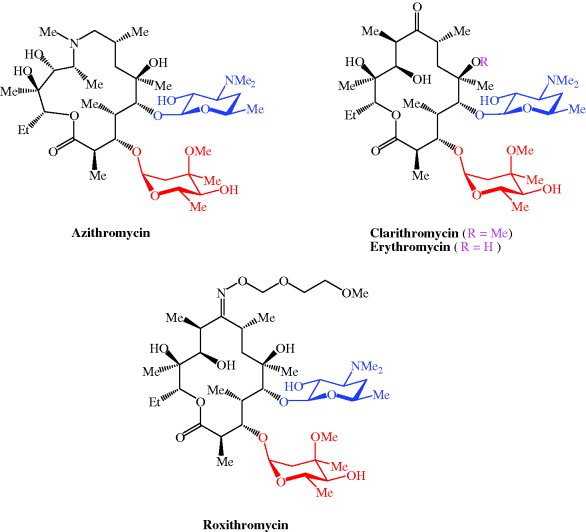
Antibiotics | Free Full-Text | From Erythromycin to Azithromycin and New Potential Ribosome-Binding Antimicrobials

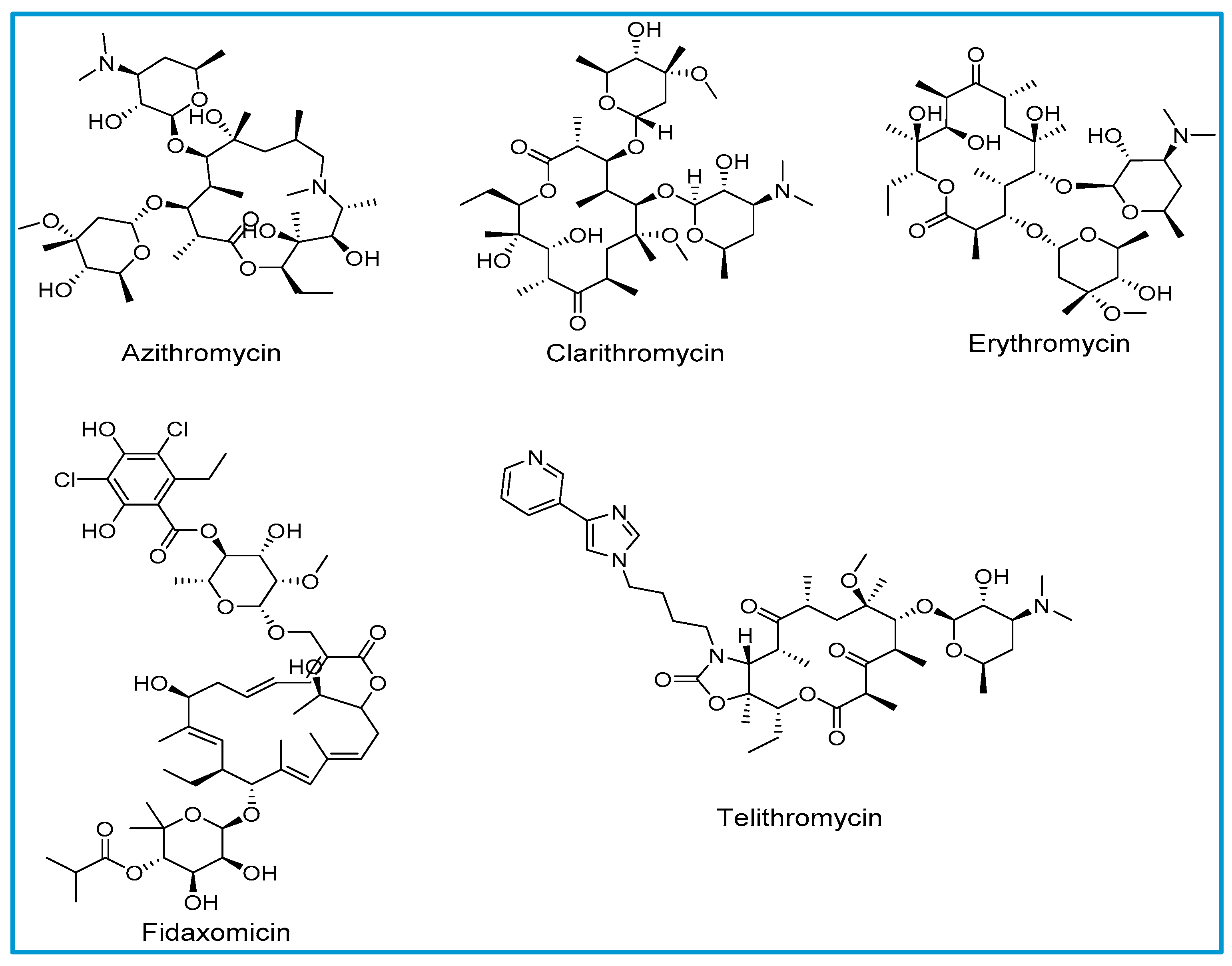
Tetracyclines, Macrolides, Clindamycin, Chloramphenicol, Streptogramins, and Oxazolidinones - Basic and Clinical Pharmacology, 13th Ed.

Macrolide Resistance Conferred by Base Substitutions in 23S rRNA | Antimicrobial Agents and Chemotherapy

Synthesis of New 14-Membered Macrolide Antibiotics via a Novel Ring Contraction Metathesis | Organic Letters

Macrolide Core Synthesis of Calysolin IX Using an Intramolecular Glycosylation Approach - Nawój - 2020 - European Journal of Organic Chemistry - Wiley Online Library
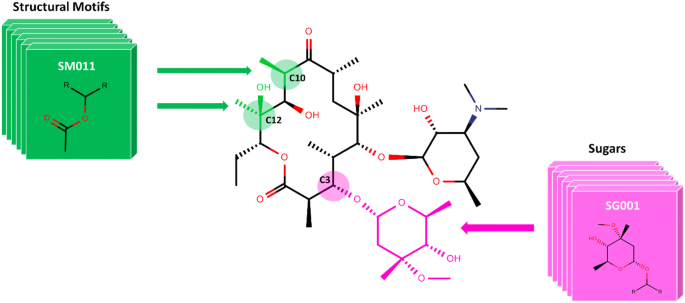
SIME: synthetic insight-based macrolide enumerator to generate the V1B library of 1 billion macrolides | Journal of Cheminformatics | Full Text

A relay ring-opening/double ring-closing metathesis strategy for the bicyclic macrolide-butenolide core structures - RSC Advances (RSC Publishing) DOI:10.1039/C4RA10937F

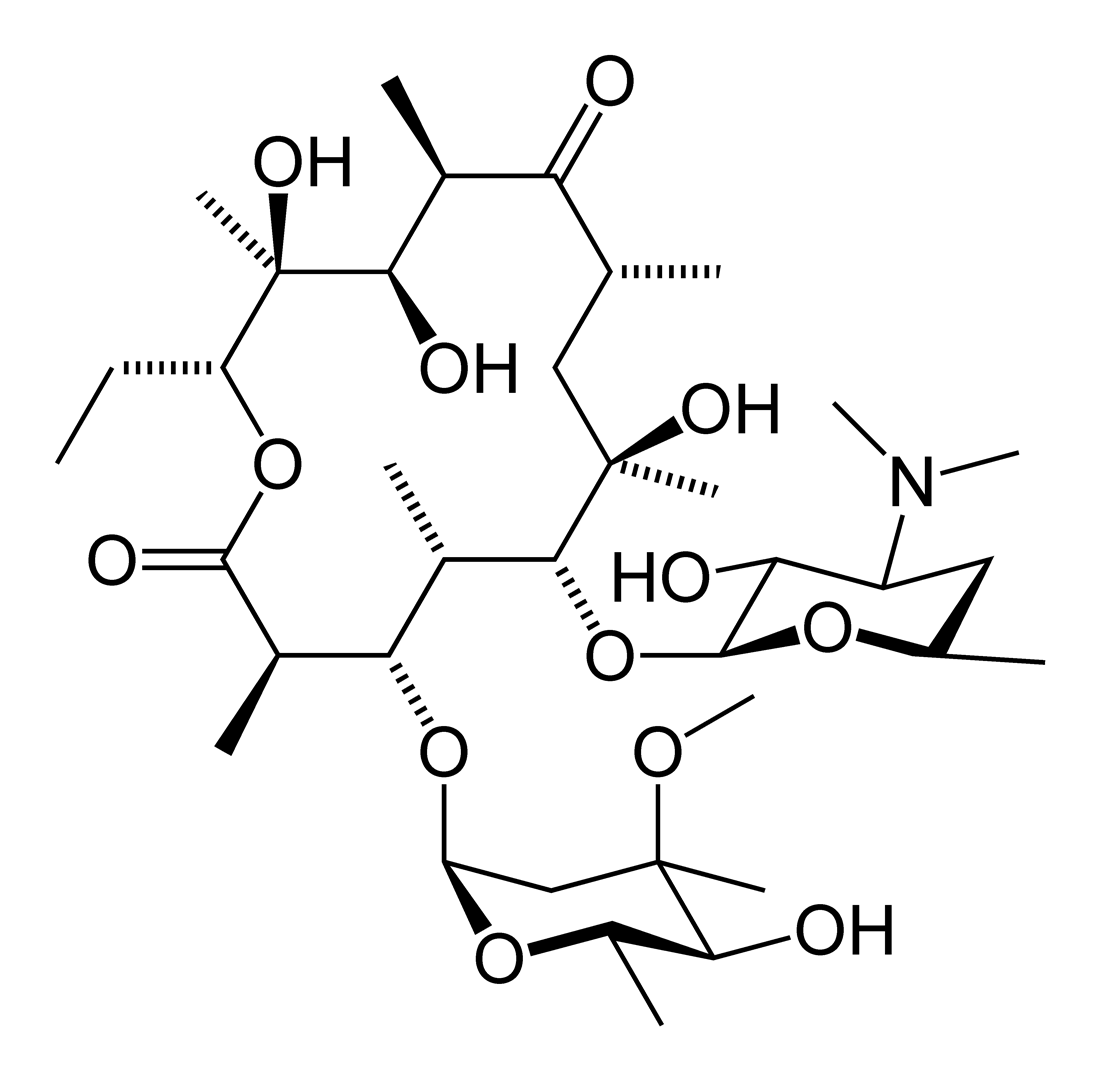
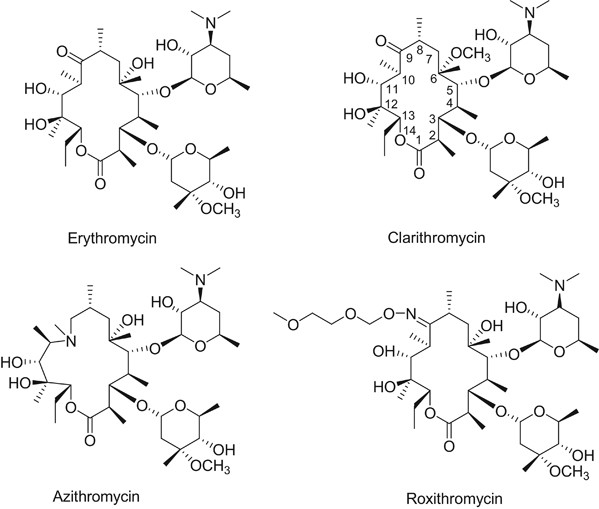
Macrolides. The macrolides are a group of closely related compounds characterized by a macrocyclic lactone ring (usually containing 14 or 16 atoms) to. - ppt download